2012
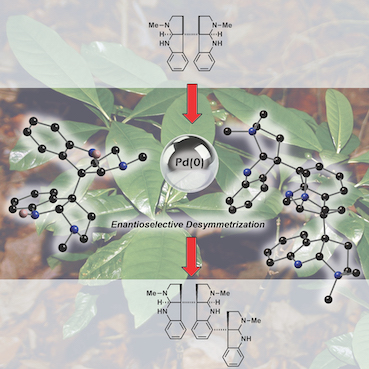
“Catalytic enantioselective desymmetrization as a tool for the synthesis of Hodgkinsine and Hodgkinsine B”, Robert H. Snell, Matthew J. Durbin, Robert L. Woodward and Michael C. Willis, Chem. – Eur. J. 2012, 18, 16754 - 16764. (doi:10.1002/chem.201203150)
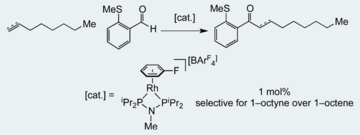
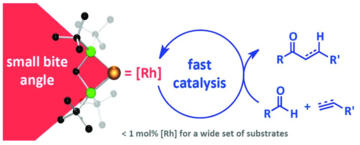
“Exploring small bite–angle ligands for the rhodium–catalyzed intermolecular hydroacylation of beta–S–substituted aldehydes with 1–octene and 1–octyne”, Indrek Pernik, Joel F. Hooper, Adrian B. Chaplin, Andrew S. Weller* and Michael C. Willis, ACS Catalysis, 2012, 2, 2779 - 2786. (doi: 10.1021/cs300541m)
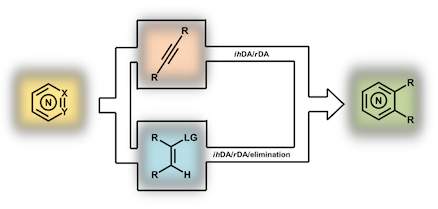
“Tandem inverse-electron-demand hetero/retro Diels-Alder reactions for aromatic nitrogen heterocycle synthesis”, Radleigh A. A. Foster and Michael C. Willis, Chem. Soc. Rev. 2013, 42, 63 - 76. (doi: 10.1039/C2CS35316D)
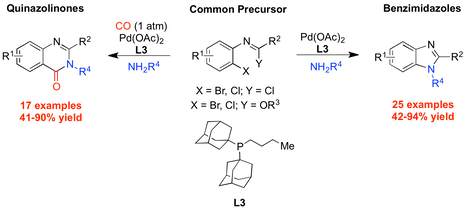
“Palladium-catalyzed synthesis of benzimidazoles and quinazolinones from common precursors”, Jessie E.R. Sadig, Radleigh Foster, Florian Wakenhut and Michael C. Willis, J. Org. Chem. 2012, 77, 9473-9486. (doi: 10.1021/jo301805d). Selected by the Editors as a “Feature Article”. SynFacts 2013, 9, 147.
“Intermolecular alkyne hydroacylation. Mechanistic insight from the isolation of the vinyl intermediate that precedes reductive elimination”, Rebekah Pawley, Miguel Huertos, Guy Lloyd–Jones, Andrew S. Weller*, Michael C. Willis, Organometallics 2012, 31, 5650-5659. (doi: 10.1021/om300647n)
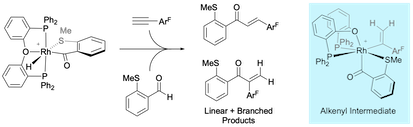
“Rhodium-catalysed linear-selective alkyne hydroacylation”, Sarah-Jane Poingdestre, Jonathan D. Goodacre, Andrew S. Weller and Michael C. Willis, Chem. Commun. 2012, 48, 6354-6356. (doi: 10.1039/C2CC32713A)

“Copper-catalyzed tandem C-N bond formation: An efficient annulative synthesis of functionalised cinnolines”, Catherine J. Ball, Jeremy Gilmore and Michael C. Willis, Angew. Chemie Int. Ed. 2012, 51, 5718-5722. (doi: 10.1002/anie.201201529). SynFacts 2012, 8, 833.
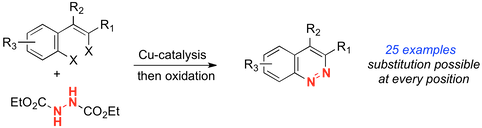
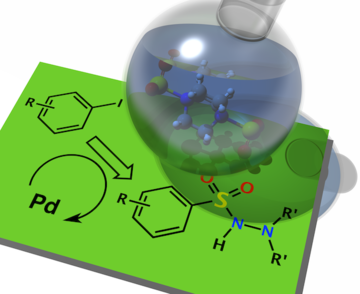
“Palladium-catalysed aminosulfonylation of aryl-, alkenyl- and heteroaryl halides: Scope of the three-component synthesis of N-aminosulfonamides”, Edward J. Emmett, Charlotte S. Richards-Taylor, Bao Nguyen, Alfonso Garcia-Rubia, Barry R. Hayter and Michael C. Willis, Org. Biomol. Chem. 2012, 10, 4007 - 4014. (doi; 10.1039/C2OB07034K). Featured on the front cover of the journal.
“Intermolecular hydroacylation: High activity rhodium catalysts containing small bite angle diphosphine ligands.” Adrian B. Chaplin, Joel F. Hooper, Andrew S. Weller and Michael C. Willis, J. Am. Chem. Soc. 2012, 134, 4885-4897. (doi: 10.1021/ja211649a)
“Palladium-catalyzed direct functionalization of benzoxazoles with alkenyl iodides”, Maria Gerelle, Anne J. Dalencon, Michael C. Willis, Tetrahedron Lett. 2012, 53, 1954-1957. (doi: 10.1016/j.tetlet.2012.02.014)

“Aryl methyl sulfides as substrates for rhodium-catalyzed alkyne carbothiolation: Arene functionalization with activating group recycling”, Joel F. Hooper, Adrian B. Chaplin, Carlos González-Rodríguez, Amber L. Thompson, Andrew S. Weller and Michael C. Willis, J. Am. Chem. Soc. 2012, 134, 2906 - 2909. (doi: 10.1021/ja2108992). SynFacts 2012, 8, 534.

“Author profile”, Angew. Chem. Int. Ed. 2012, 51, 1304. (doi: 10.1002/anie.201107516).


